Anatomical variations in right colon vascularization:
Its implication in complete mesocolic excision with D3
lymphadenectomy. A cross-sectional study.
Juan Pablo Campana1, Catalina Poggi1,
Lorena Savluk2, José Viñas1,
Esteban González Salazar1, Ricardo Mentz1,
Gustavo Rossi1,
Sergey Efetov3,4, Kirill Puzakov4,
Albina Zubayraeva4, Carlos Alberto Vaccaro1
1 Colorectal Surgery
Section, General Surgery Department, Hospital Italiano de Buenos Aires,
Argentina.
2 Imaging Diagnostic Service, Hospital Italiano de
Buenos Aires, Argentina.
3 Surgical Department N2, University Clinical Hospital
N4
4 I.M. Sechenov First Moscow State Medical University
ABSTRACT
Background:
Complete mesocolic excision with D3 lymphadenectomy (CME-D3) improves the
outcomes of patients operated on for colon cancer. Proper recognition of
vascular anatomy is essential to avoid complications.
Aim: Primary
outcome was to determine the prevalence of anatomical variations of the
superior mesenteric artery (SMA) and its branches in relation to the superior
mesenteric vein (SMV). Secondary outcome was to evaluate the association
between these anatomical variations and sex and ethnicity of the patients.
Design:
Cross-sectional study.
Material
and methods: Two hundred twenty-five patients with right colon cancer
diagnosed between January 2017 and December 2020 were included. Two independent
radiologists described the vascular anatomy of computed tomography angiography
scans. The population was divided into 2 groups and subdivided into 6 groups
(1a–c, 2a–c), according to the relationship of the SMA and its branches with
the SMV.
Results:
The ileocolic artery was constant, crossing the SMV on its posterior aspect
in 58.7% of the cases. The right colic artery, present in 39.6% of the
patients, crossed the SMV on its anterior aspect in 95.5% of the cases. The
most frequent subgroup variant was 2a followed by 1a (36.4 and 24%,
respectively). No association was found between anatomical variants and gender
or ethnic origin.
Conclusions:
The anatomical variations of the SMA and its branches are common, with no
predominant pattern. There was no association between anatomical variations and
gender or ethnic origin in our cohort. Preoperative evaluation of these
variations by computed tomography angiography scan is useful to avoid vascular
injuries during CME-D3.
Key
words: complete mesocolic excision, D3 lymphadenectomy, vascular anatomy,
right colon
The authors
declare no conflicts of interest.
Correspondence: Juan Pablo
Campana. juan.campana@hospitalitaliano.org.ar. Tte. Gral. Juan Domingo Perón 4190, CABA. CP
C1199ABB. Ph: (+54 11) 4959–0200.
Juan Pablo
Campana ORCID 0000-0002-0420-5906; Catalina
Poggi ORCID 0000-0002-3430-8375
Lorena Savluk ORCID 0000-0002-9939-8260; José Viñas ORCID 0000-0001-8615-7436
Esteban
González Salazar ORCID 0000-0003-3831-522X; Ricardo
E. Mentz ORCID 0000-0002-6746-8869
Gustavo L.
Rossi 0000-0002-4451-6709; Sergey Efetov 0000-0003-0283-2217
Kirill Puzakov ORCID 0000-0001-9017-8205; Albina Zubayraeva ORCID 0000-0001-8284-3922;
Carlos A.
Vaccaro ORCID 0000-0002-1299-5864
INTRODUCTION
Extrapolated
from oncologic surgery for rectal cancer, the concept of complete mesocolic
excision (CME) with central vascular ligation and D3 lymphadenectomy in right
hemicolectomy for colon cancer has recently re-emerged.1 Compared to
standard mesocolic resection (conventional D2 right hemicolectomy), this
surgical strategy presents a greater 5-year overall survival and a greater
number of lymph nodes removed, although it could potentially be associated with
a greater number of complications.2-5
In
oncological resection of the right colon, D3 lymphadenectomy corresponds to the
dissection of the central lymph node groups located on the anterior face of the
superior mesenteric vein (SMV) from the level of the middle colic vein (MCV) to
the ileocolic vein (IVC).6 To perform this dissection it is
essential to know the vascular anatomy of the right colon, including its
anatomical variations, to minimize the possibility of intraoperative vascular
accidents. In particular, it is important to know the relationships of the
arterial branches with the SMV and its branches, since lymphadenectomy may be
more difficult when the ileocolic artery (ICA) crosses posterior to the SMV.7
Currently,
there are few published studies that relate the vascular anatomy of the right
colon to D3 dissection and that describe the most frequent relationships of the
SMA and its branches with the SMV.8,9
The
main objective of this study was to determine the prevalence of the different
anatomical variations of the SMA and its branches and their relationships with
the SMV studied by computed tomography (CT) in patients with undergoing right
hemicolectomy for colon cancer in a closed cohort from Argentina.
Secondly,
it was proposed to evaluate the association between the different anatomical
variants with the sex and ethnicity of the patients.
MATERIAL
AND METHODS
An
observational cross-sectional study was carried out that included patients
older than 18 years with cancer of the right colon who underwent surgery
consecutively in a tertiary hospital in Argentina between January 2017 and
December 2020.
Cancer
of the right colon was defined as adenocarcinoma located in the cecum,
ascending colon, hepatic flexure, and proximal transverse colon.
All
patients underwent a CT to study the different anatomical variations of the SMA
and its branches and their relationships with the SMV.
The
excluded patients were those: 1) with previous major abdominal resections by
laparotomy or laparoscopy, 2) whose preoperative CT was without intravenous
contrast, and 3) in whom both radiologists did not agree on the definitive
vascular diagnosis.
The
ethnic origin was divided into 4 groups: European, Amerindian, African and
Asian, according to the subcontinental origin of the ancestry components of the
different individuals.10
The
study was approved by the Ethics Committee for Institutional Research Protocols
(CEPI) of the Hospital Italiano de Buenos Aires.
Tomographic protocol
The tube
current was adjusted by automatic exposure control based on the patient's body
constitution (300-550 mA/s). The tube power was 120 kVp and
the slice
thickness was 0.5-1.0 mm. The
non-ionic and iodinated contrast Omnipaque® (350 mg Iodine/ml injectable
solution) was used. Contrast
was administered at 3.0-3.5 ml at a dose of 1-2 ml/kg body weight. Studies were
reported according to the following protocol: non-contrast phase; arterial phase (the scan starts
automatically after maximum contrast enhancement in the aorta); venous phase (imaging delayed 40-50
seconds after bolus injection); late phase (10 minutes after bolus injection) for evaluation of
the urinary tract.
To assess the
vascular anatomy in detail, 3D volume rendering (3D reconstruction) was used as
exemplified in Fig. 1.

Figure
1. Computed tomography angiography
with 3D reconstruction. The ileocolic artery (AIC) can be seen crossing the
superior mesenteric vein (VMS) on its anterior face aspect, and the right colic
artery (ACD) crossing it on its posterior aspect. It corresponds to the type 1c
variant.
Definitions
The
middle colic artery (MCA), defined as the first branch of the SMA, goes to the
transverse colon through the mesocolon. The ICA was defined as the last branch
that emerges from the right side of the SMA and approaches the ileocecal valve.
The right colic artery (RCA) was defined as that branch that arises from the
SMA between the MCA and the ICA, in the direction of the ascending colon. This
vessel can also originate from a common trunk with the MCA or ICA.
Based
on the classification by Efetov et al.,11 we divided the population
into two groups according to the
relationships
between ICA and SMV: Type 1: ICA runs ventrally to the SMV. Type 2: ICA runs
dorsally to the SMV.
Taking
into account the relationships of the MCA and RCA with the SMV, six possible
scenarios emerge, schematized in Fig. 2:
Type
1a: both ICA and MCA run ventrally to the SMV and RCA is absent.
Type
1b: ICA, RCA, and MCA run ventrally to the MSV. Type 1c: ICA and MCA run ventrally
to the SMV, while RCA run dorsally.
Type
2a: ICA runs dorsally to the SMV, MCA runs ventrally, and RCA is absent.
Type
2b: ICA and RCA lie dorsally to the SMV and MCA lies ventrally. Type 2c: ICA
runs dorsally to the SMV, while
RCA
and MCA run ventrally.

Figure 2. Classification of anatomical variants. A) Type 1a. B) Type 1b. C) Type
1c. D) Type 2a. E) Type 2b. F) Type 2c. VMS: superior mesenteric vein. SMA:
superior mesenteric artery. MCA: middle colic artery. ICA: ileocolic artery.
RCA: right colic artery.
Procedure
All
CTs were independently assessed by two radiologists who described the vascular
anatomy as previously defined. To minimize the bias related to
interobservational variation, a description of the vascular anatomy was
performed according to standardized criteria.11
Sample
calculation
A
simple consecutive sampling method was carried out. The calculation of the
sample size was made based on the Argentine population according to the 2022
census. Taking into account 70% of the population over 18 years of age (according
to census data) and considering a margin of error of 6% with a 95% confidence
level, the calculated sample size was 225 patients.
Statistic
analysis
Categorical
variables were reported as proportions and continuous variables as means with
their respective standard deviations or medians with their interquartile
ranges, according to distribution. The chi-square test or Fisher's exact test
was used for categorical variables and Student's t-test for continuous
variables. All the statistical tests were two-tailed and a value of p<0.05
was considered statistically significant. STATA®, version 13 software
(StataCorp LLC, Texas, United States) was used for all analyses.
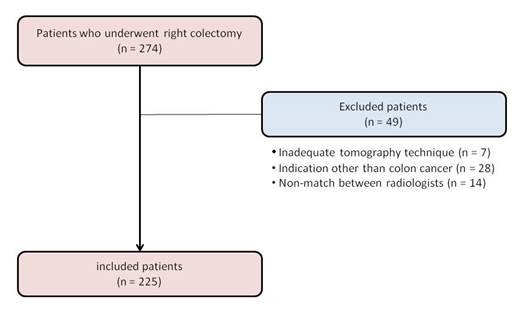
RESULTS
Of
the 274 patients operated on, 49 were excluded, leaving a total of 225 patients
for analysis (Fig. 3).
The
sex of the population was predominantly female (60%) and the
mean
age was 74±12 (range: 33-94) years. The most frequent tumor locations were the
cecum (44%) and ascending colon (43.1%). The most prevalent ethnic group was
European (97.8%). The rest of the demographic and clinical characteristics are
presented in Table 1.
ICA
was present in all patients. In 58.7% of the patients (n=132) it ran dorsally
to the SMV (Type 2). RCA was present in 39.6% of patients (n=89); it was a
branch of the MCA in 47.2% of the cases, a direct branch of the SMA in 45.8%
and a branch of the ICA in only 7%. The RCA run ventrally to the SMV in 95.5%
of the patients (n=85).
The
MCA was present in 98.7% of the patients
(n=222); in all of them it emerged from the anterior aspect of the SMA. Only
one patient presented, as an anatomical variation, a common trunk between the
ICA and the MCA, in which case the RCA was absent.
Table
1. Demographic variables and clinical characteristics.
|
Variables
|
Patients
n = 225
|
|
|
|
Age, mean ± SD
|
74 ± 12
|
|
|
Women % (n)
|
60 (135)
|
|
|
Ethnicity, % (n)
European
Amerindian
African
Asian
|
97.8 (220)
0.4 (1)
0
1.8 (4)
|
|
|
Weight, mean ± SD
|
71 ± 17
|
|
|
Height, mean ± SD
|
1.6 2 ± 0.1
|
|
|
BMI, mean ± SD
|
27 ± 5
|
|
|
Location, % (n)
Cecum
Ascending colon
Hepatic flexure
Transverse colon
|
44 (99)
43.1 (97)
8 (18)
4.9 (11)
|
|
SD:
standard deviation. BMI: body mass index.
The
most frequent subgroup variant was type 2a followed by type 1a (36.4 and 24%, respectively).
The prevalence of the different combinations of variations is presented in
Table 2.
Table 2. Frequency
of anatomical variations.
|
Anatomical variations
|
Patients
n = 225
|
|
|
|
ICA present, % (n)
|
100 (225)
|
|
|
RCA present, % (n)
|
39.6 (89)
|
|
|
MCA present, % (n)
|
98.7 (222)
|
|
|
Type 1, % (n)
1a
1b
1c
|
41.3 (93)
24 (54)
17.3 (39)
0
|
|
|
Type 2, % (n)
2a
2b
2c
|
58.7 (132)
36.4 (82)
1.8 (4)
20.4 (46)
|
|
ICA: ileocolic
artery; RCA: right colic artery; MCA: middle colic artery.
The
course of the ICA along the anterior aspect of the SMV vs. the posterior aspect
was not associated with the patient gender (women: 61.3 vs. 59.1%,
respectively; p=0.74) or the ethnic origin (European: 98.9 vs. 97%, Amerindian:
0 vs. 0.8%, African: 0 vs. 0%, Asian: 1.1 vs. 2.3%, respectively; p=0.56)
(Table 3).
There
was also no association between the presence of RCA and gender (women: 56.2 vs.
62.5%; respectively; p=0.34) or ethnicity (European: 96.6 vs. 98.5%, Amerindian:
1.1 vs. 0%, African: 0 vs. 0%, Asian: 2.2 vs. 1.5%, respectively; p=0.42)
(Table 4).
Table
3. Bivariate analysis according to the ileocolic artery.
|
Variables
|
Type 1 (n = 93)
|
Type 2 (n = 132)
|
P value
|
|
|
|
Age, mean ± SD
|
73 ± 12
|
75 ± 12
|
0.38
|
|
|
Women, % (n)
|
61.3 (57)
|
59.1 (78)
|
0.74
|
|
|
Ethnicity, % (n)
European
Amerindian
African
Asian
|
98.9 (92)
0
0
1.1 (1)
|
97 (128)
0.8 (1)
0
2.3 (3)
|
0.56
|
|
|
Weight, mean ± SD
|
71 ± 16
|
71 ± 17
|
0.98
|
|
|
Height, mean ± SD
|
1.62 ± 0.1
|
1.61 ± 0.1
|
0.42
|
|
|
BMI, mean ± SD
|
26 ± 4
|
27 ± 5
|
0.62
|
|
|
Location, % (n)
Cecum
Ascending colon
Hepatic flexure
Transverse colon
|
39.8 (37)
46.2 (43)
9.7 (9)
4.3 (4)
|
47 (62)
40.9 (54)
6.8 (9)
5.3 (7)
|
0.64
|
|
SD: standard deviation. BMI: body mass
index.
DISCUSSION
To
the best of our knowledge and experience, this descriptive study represents the
first to analyze the vascular anatomy of the right colon in a Latin American
population using CT scan with IV contrast. In this population, the ICA is found
more frequently on the posterior aspect of the SMV. The RCA was present in
approximately half of the patients and in most cases it crossed the SMV on its
anterior aspect. In the two cases in which the RCA passed through the posterior
aspect of the SMV, the ICA did so in the same way. This coincides with the
findings reported by Murono et al.12
The
vascular variations of the SMA and its branches have been addressed in the
literature from an anatomical point of view. Most of the studies are
non-systematic reviews13 or descriptive cadaveric studies.6,14,15
However, none of them emphasize the anatomical relationships of the arterial
branches supplying the colon with the SMV, which is the most important
technical landmark when performing CME with central ligation and D3 lymphadenectomy.
Table
4. Bivariate analysis according to the right colic artery.
|
Variables
|
RCA present
(n = 89)
|
RCA absent
(n = 136)
|
P value
|
|
|
|
Age, mean ± SD
|
75 ± 11
|
73 ± 12
|
0.22
|
|
|
Women, % (n)
|
56.2 (50)
|
62.5 (85)
|
0.34
|
|
|
Ethnicity, % (n)
European
Amerindian
African
Asian
|
96.6 (86)
1.1 (1)
0
2.2 (2)
|
98.5 (134)
0
0
1.5 (2)
|
0.42
|
|
|
Weight, mean ± SD
|
72 ± 14
|
70 ± 19
|
0.6
|
|
|
Height, mean ± SD
|
1.62 ± 0.1
|
1.61 ± 0.1
|
0.2
|
|
|
BMI, mean ± SD
|
27 ± 4
|
27 ± 5
|
0.96
|
|
|
Location, % (n)
Cecum
Ascending colon
Hepatic flexure
Transverse colon
|
42.7 (38)
47.2 (42)
6.7 (6)
3.4 (3)
|
44.9 (61)
40.4 (55)
8.8 (12)
5.9 (8)
|
0.68
|
|
RCA: right colic artery. SD:
standard deviation. BMI: body mass index.
The
ICA is a constant branch of the SMA. The literature disagrees regarding on
which is the most frequent relationship between the ICA and the SMV. Consistent
with the findings of some studies,14-16 in our population the ICA is
found more frequently (58.7%) posterior to the SMV.
On
the other hand, the RCA is much more variable. While most groups report that it
is present in approximately 30% of cases17 and is located anterior
to the SMV, in our population its presence slightly exceeded this number
(36.9%), also crossing mostly by the anterior aspect of the SMV.
Regarding
the MCA, in our series it was present in 98.7% of the patients, always emerging
from the anterior aspect of the SMV, in accordance with other reports.13,17
The
most frequent combinations of anatomical variations in our study were types 2a
and 1a (36.4 and 24%, respectively). Likewise, in the Russian population type
2a predominated with 43.8% of cases. In contrast, in the Chinese population
type 1a predominates with 30.8% and the position of the ICA with respect to
the SMV is balanced,11 while in more than 60% of our patients the
ICA crosses the SMV by its posterior aspect.
Our
population is highly influenced by European migrations,18 which
could explain our findings, which are more similar to those of the Russian
population. In our series, European ancestry was higher than the rest of the
subgroups (97.8%).
We found no association between the course of the ICA and the
gender or ethnic origin of each individual. We also did not detect an
association between the presence of the RCA and these same variables.
Therefore, the anatomical differences published between the eastern and western
population could not be confirmed in our study. However, we believe this could
be because the sample size calculation was performed to assess prevalence and
not subgroups differences. Future studies should expand the sample to determine
differences between different ethnic groups.
The
variability of the vascular anatomy of the right colon and that of the
relationships between the branches of the SMA and SVM offer an additional
difficulty for the surgeon. Currently, it is feasible to know the anatomy
through preoperative studies, being the CT scan with IV contrast and 3D
vascular reconstruction a highly specific and sensitive non-invasive study for
its evaluation.19,20 The preoperative assessment of these studies
should become a regular practice by the surgical team in order to know the
vascular anatomy that will have to face during surgery and thus reduce the most
important intraoperative morbidity related to D3 lymphadenectomy, such as SMV
injury.21
This
study has some limitations that deserve to be highlighted. First, the
cross-sectional design carries the limitation of the interpretation of
causality for association analyses. However, in our study the associations were
only evaluated in variables unalterable over time, such as anatomical
variations, gender and ethnic traits. Secondly, the sampling was of a simple
consecutive type, which facilitates the incorporation of individuals into the
study but has the drawback of being less representative of the general
population. Third, the different anatomical variants were evaluated by CT scan
and not confirmed during the surgical procedure, which may decrease the quality
of the data collected. However, CT scan with 3D reconstruction has shown high
sensitivity and specificity for the diagnosis of vascular anatomy in numerous
publications.12,22 In addition, all CT scans were analyzed by two
independent radiologists and only those studies in which both professionals
agreed on the diagnosis of vascular anatomy were included.
CONCLUSION
The
SMA and its branches have variable relationships with the SMV, without a
predominant pattern.
There
was no association between these variations and the gender or ethnic origin of
the population analyzed.
Evaluation
of these anatomic variants by CT angiography should be routinely incorporated
into the preoperative planning of a right colectomy with CME and D3
lymphadenectomy, in order to avoid undesirable intraoperative complications.
REFERENCES
1. Hohenberger W, Weber
K, Matzel K, Papadopoulos T, Merkel S. Standardized surgery for colonic cancer:
complete mesocolic excision and central ligation-technical notes and outcome.
Colorectal Dis. 2009; 11:354-64; discussion 364-65.
2.
West NP,
Kobayashi H, Takahashi K, Perrakis A, Weber K, Hohenberger W, et al.
Understanding optimal colonic cancer surgery: comparison of Japanese D3
resection and European complete mesocolic excision with central vascular
ligation. J Clin Oncol. 2012; 30:1763-69.
3.
Kanemitsu
Y, Komori K, Kimura K, Kato T. D3 lymph node dissection in right hemicolectomy
with a no-touch isolation technique in patients with colon cancer. Dis Colon
Rectum. 2013; 56:815-24.
4.
Bertelsen
CA, Neuenschwander AU, Jansen JE, Tenma JR, Wilhelmsen M, Kirkegaard-Klitbo A,
et al. 5-year outcome after complete mesocolic excision for right-sided colon
cancer: a population-based cohort study. Lancet Oncol. 2019; 20:1556-65.
5.
Gao Z,
Wang C, Cui Y, Shen Z, Jiang K, Shen D, et al. Efficacy and safety of complete
mesocolic excision in patients with colon cancer: three-year results from a
prospective, nonrandomized, double-blind, controlled trial. Ann Surg. 2020; 271:519-26.
6.
Spasojevic
M, Stimec BV, Dyrbekk APH, Tepavcevic Z, Edwin B, Bakka A, et al. Lymph node
distribution in the D3 area of the right mesocolon: implications for an
anatomically correct cancer resection. A postmortem study. Dis Colon Rectum.
2013; 56:1381-87.
7. Ishiyama Y, Maeda C, Shimada S, Kudo
SE. Propensity-score-matched analysis of short- and long-term outcomes in
patients with an ileocolic artery crossing anterior vs posterior to the
superior mesenteric vein during curative resection for right-sided colon cancer.
Surg Endosc. 2020; 34:5384-92.
8.
Willard
CD, Kjaestad E, Stimec BV, Edwin B, Ignjatovic D, RCC Study Group. Preoperative
anatomical road mapping reduces variability of operating time, estimated blood
loss, and lymph node yield in right colectomy with extended D3 mesenterectomy
for cancer. Int J Colorectal Dis. 2019; 34:151-60.
9.
Wu C, Ye
K, Wu Y, Chen Q, Xu J, Lin J, et al. Variations in right colic vascular anatomy
observed during laparoscopic right colectomy. World J Surg Oncol. 2019; 17:16.
10. Homburger JR, Moreno-Estrada A, Gignoux CR,
Nelson D, Sanchez E, Ortiz-Tello P, et al. Genomic insights into
the ancestry and demographic history of South America. PLoS Genet. 2015; 11:e1005602.
11. Efetov S, Jiang J,
Liu Z, Tulina I, Kim V, Schegelski V, et al. Superior mesenteric vessel anatomy
features differ in Russian and Chinese patients with right colon cancer:
computed tomography-based study. Chin Med J. 2021; 134:2495-97.
12. Murono K, Kawai K, Ishihara S, Otani
K, Yasuda K, Nishikawa T, et al. Evaluation of the vascular anatomy of the
right-sided colon using three-dimensional computed tomography angiography: a
single-center study of 536 patients and a review of the literature. Int J Colorectal
Dis. 2016; 31:1633-38.
13. Alsabilah J, Kim WR,
Kim NK. Vascular structures of the right colon: incidence and variations with
their clinical implications. Scand J Surg. 2017; 106:107-15.
14.
Kuzu MA,
Ismail E, Çelik S, Şahin MF, Güner MA, Hohenberger W, et al. variations in the
vascular anatomy of the right colon and implications for right-sided colon
surgery. Dis Colon Rectum. 2017; 60: 290-98.
15.
Shatari T, Fujita M,
Nozawa K, Haku K, Niimi M, Ikeda Y, et al. Vascular anatomy for
right colon lymphadenectomy. Surg Radiol Anat. 2003; 25:86-88.
16. Ignjatovic D, Sund S,
Stimec B, Bergamaschi R. Vascular relationships in right colectomy for cancer:
clinical implications Tech Coloproctol. 2007; 11:247-50.
17.
Lee SJ,
Park SC, Kim MJ, Sohn DK, Oh JH. Vascular anatomy in laparoscopic colectomy for
right colon cancer. Dis Colon Rectum. 2016; 59:718-24.
18. Muzzio M, Motti JMB,
Paz Sepulveda PB, Yee MC, Cooke T, Santos MR, et al. Population structure in
Argentina. PLoS One. 2018; 13:e0196325.
19. Miyamoto R, Tadano S,
Sano N, Inagawa S, Adachi S, Yamamoto M. The impact of three-dimensional
reconstruction on laparoscopic-assisted surgery for right-sided colon cancer.
Wideochir Inne Tech Maloinwazyjne. 2017; 12:251-56.
20. Shioyama Y, Kimura M, Horihata K,
Masuda M, Hagihira T, Okumura T, et al. Peripancreatic
arteries in thin-section multislice helical CT. Abdom Imaging. 2001; 26:234-42.
21. Bertelsen CA, Neuenschwander AU,
Jansen JE, Kirkegaard-Klitbo A, Tenma JR, Wilhelmsen M, et al. Short-term
outcomes after complete mesocolic excision compared with “conventional” colonic
cancer surgery. Br J Surg. 2016; 103:581-89.
22. Coffey JC. Commentary on “Navigating
the mesentery: a comparative pre- and per-operative visualization of the
vascular anatomy.” Colorectal Dis. 2015; 17:818-19.


